BEBPA Blog
Tech Briefing: Overview of Recent FDA Guidance Document for the Validation of Clinical Biomarker Assays
By Laureen Little, President, Quality Aspects & BEBPA
A clinically validated biomarker can significantly influence clinical study timelines. Biomarkers may be used at multiple stages of a study, including disease detection, diagnosis, prognosis, prediction of treatment response, and disease monitoring. Another useful way to consider biomarkers is in the context of disease progression
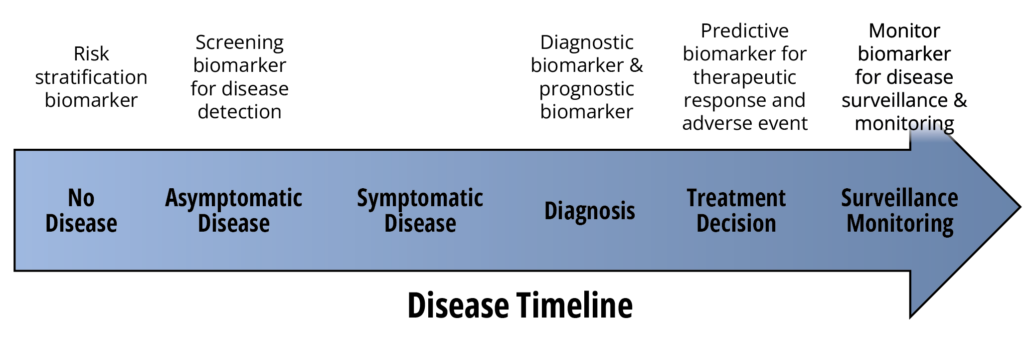
Unfortunately, this approach can create confusion between clinical and analytical development teams because clinical validation and analytical validation are distinct processes. This distinction becomes clearer with more precise wording. Clinical validation demonstrates that a measurable biomarker is a relevant indicator of normal biological processes, disease processes, or biological responses to an exposure or intervention, including treatment. Once a biomarker is identified, an analytical method must be developed to measure it. That method must then undergo analytical validation before its results can be submitted to regulatory authorities in support of product approval.
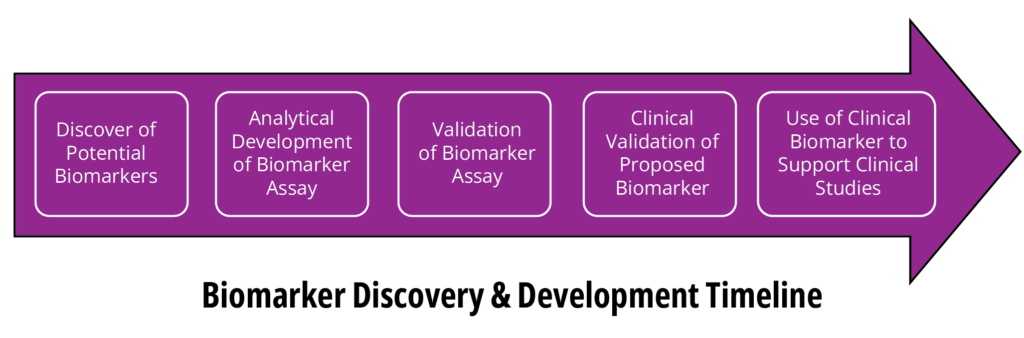
A major gap in scientific discussions about biomarkers has been the lack of practical guidance on how to analytically validate biomarker assays. In April 2026, the FDA issued its guidance, “Bioanalytical Method Validation for Biomarkers Guidance for Industry” (Reference 2). However, this document provides far less implementation detail than other analytical method validation guidances (References 3 and 4). Its most useful direction is the following:
“Method validation for biomarker assays should address the same questions as method validation for drug assays. The accuracy, precision, sensitivity, selectivity, parallelism, range, reproducibility, and stability of a biomarker assay are important characteristics that define the method. The approach described in the guidance for industry M10 Bioanalytical Method Validation and Study Sample Analysis (November 2022) for drug assays should be the starting point for validation of biomarker assays, especially chromatography and ligand-binding based assays.”
It goes on to state that the following key questions should asked when developing a biomarker validation:
- Does the method measure the intended analyte? For example, does anything interfere with the measurement, and is the method specific or selective for the analyte?
- What is the variability associated with these measurements? For example, what are the accuracy and precision of the method?
- What is the range in measurements that provide reliable data? For example, what is the sensitivity of the method (e.g., what is the lower limit of quantitation of the method (LLOQ) and upper limit of quantitation of the method (ULOQ))?
- How do sample collection, handling, and storage affect the reliability of the data from the bioanalytical method? For example, what steps need to be followed while collecting samples? Do the samples need to be frozen during shipping? What temperatures are required to store the samples, and how long can the samples be stored? How are samples affected by benchtop manipulations?
The guidance offers little detail on key validation characteristics such as precision (including interassay precision, repeatability, and robustness), accuracy, linearity, and interfering substances. It does not provide definitions or suggest the number of samples/runs needed. Nor does it address approaches for different matrices or potential sources of interference. Part of the challengeof writing such a document is the wide variability among biomarker types, which makes it difficult to provide specific recommendations. Given this limited guidance, benchmarking across the industry is essential to understand current practices in biomarker analytical development and validation.
BEBPA is launching its first virtual conference on this topic and has lined up several industry, institutional and FDA speakers to discuss the development, validation and automation of biomarker assays. The talks include:

Bridging Discovery and Diagnostics: Early Biomarker Strategies Using the Example of Checkpoint Inhibitors
Julia Schueler
Charles River Laboratories

Statistical Considerations for Clinical Bioassays
Ivan Ordonez
Sanofi

Harmonization of Inflammatory Biomarker Assay Results using Common Reference Materials to Support Drug Development
Enrique Dalmasso
USP

Leveraging Automated Immunoassay Platforms for Rapid Cytokine Assessment in Early Drug Development
Daniel Thwaites
Labcorp

Evolution of Industry Views on Assessment of Immunogenicity of Biotherapeutics
Boris Gorovits
Regeneron Pharmaceuticals, Inc.

From Sample to Signal: A One-Pot CRISPR Assay for Malaria Detection
Heather Painter
US FDA

Establishing Analytical Rigor in Clinical Flow Cytometry: Instrument Equivalence, QC Donor Strategy, and ICS Method Transfer
Ane Ogbe
Moderna Therapeutics

Assessment of Surrogate Platforms for the Evaluation of Antibody Responses Against Highly Pathogenic Influenza Viruses
Meleri Jones
UKHSA

Coming out of the Shell: Building Scalable Influenza Assays for both Egg- and Cell-Based Vaccines
Stephen Crooke
Sanofi
Spatially Resolved System Level Biomarker Framework for Tumor Immunotherapy Development
Speaker To Be Announced
Fit-for-Purpose Biomarker Assays for Infectious Disease: Development, Validation, and Clinical Sample Readiness
Speaker To Be Announced
We have plenty of discussion time for Questions and Answers. Come listen and discuss with your scientific peers and regulatory colleagues about a wide range of problems you are dealing with now. Just starting the development of your biomarker assay? Selecting appropriate assay platforms? Thinking about automating your assay? Struggling with the implementation of a reference material to allow comparison across sites? We cover all this and more with our case studies.
Sign up soon and reserve your spot now for our June 23-24, 2026 Biomarker Conference.
