BEBPA Blog
Volume 2, Issue 7
Unlocking the Multifaceted Mechanisms of Antibody–Drug Conjugates
By Julia Gilden & Kai Hillman, Promega Corporation
Antibody–drug conjugates (ADCs) represent a promising frontier in targeted cancer therapy, marrying the specificity of monoclonal antibodies with the potent cell-killing capabilities of cytotoxic drugs. As complex bioconjugates, ADCs use multifaceted and multistep mechanisms of action (MOA) to deliver their therapeutic payloads precisely to tumor cells. In recent years, the clinical landscape has witnessed an accelerated development and approval of ADCs, reflecting both their innovative design and the expanding understanding of their molecular intricacies. Approved ADCs deploy a variety of MOAs—including direct cytotoxicity, bystander killing, receptor blockade, and internalization—while also engaging immune-mediated processes such as antibody-dependent cellular cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP), and complement-dependent cytotoxicity (CDC) (1). Looking ahead, next-generation payloads aim to broaden this therapeutic arsenal by incorporating immune modulation, altering protein expression, and directly targeting specific oncogenic mutations.
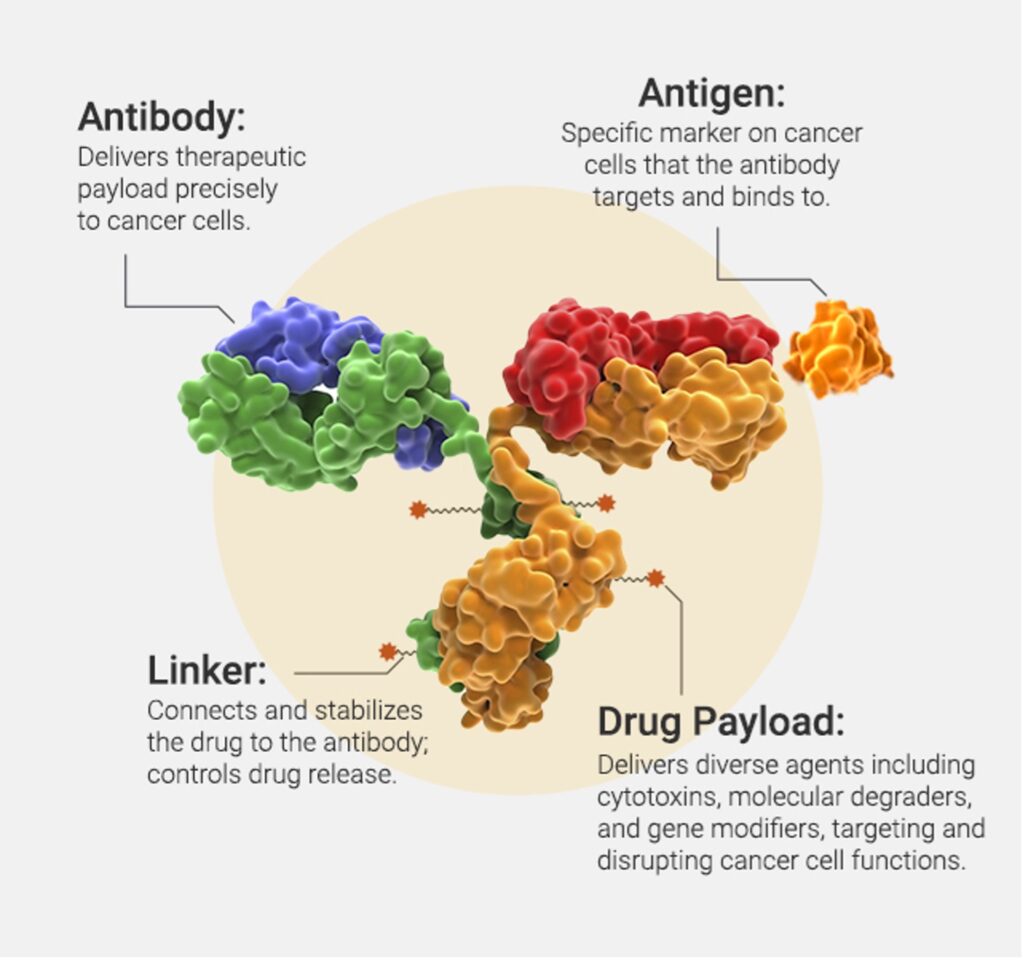
At its core, an ADC is composed of three critical components: the antibody, the linker, and the payload. The antibody confers target specificity, binding with high affinity to tumor-associated antigens, thereby ensuring that the cytotoxic drug is delivered selectively to cancer cells. The linker, which bridges the antibody and the payload, is engineered to be stable in circulation yet labile enough to release the drug once internalized by the target cell. This balance in linker design is pivotal for optimizing therapeutic efficacy while minimizing off-target toxicity. Finally, the payload itself is typically a highly potent cytotoxic agent, capable of inducing cell death even at very low concentrations, a necessity given the limited number of molecules delivered per antibody (Figure 1).
Beyond acting as delivery vehicles, the antibodies in ADCs play an active role in modulating immune responses through their Fc regions. The Fc domain can engage immune effector cells by binding to Fc receptors on natural killer cells, macrophages, and other components of the immune system. The majority of ADCs use IgG1 antibodies, due to their long serum half-life and robust Fc effector functions. These effector functions can include ADCC, ADCP, and CDC, thereby contributing an additional layer of antitumor activity. The immune-mediated functions not only assist in the direct killing of tumor cells but also help in shaping the tumor microenvironment to favor an immune response against cancer. As such, modifications in the Fc region are being explored to fine-tune these interactions and enhance overall therapeutic outcomes (2). Just like unconjugated monoclonal antibody drugs, regulators will in most cases require the Fc function of ADCs to be evaluated. Published studies of some drugs have shown that payload conjugation has little impact on Fc function (3), but as conjugation chemistry evolves, sometimes enabling very large drug-antibody-ratios (DAR), it is essential to quantify to what extent Fc function has been preserved in the final drug substance. Assays for Fc function of monoclonal antibodies, including reporter bioassays and primary cell assays, are commonly used for ADC characterization as well.
For an ADC to exert its direct cytotoxic effects, it must be internalized by the tumor cell. This process begins with the binding of the ADC to its target antigen on the cell surface, triggering receptor-mediated endocytosis. Once inside the cell, the ADC is trafficked through endosomal compartments where the linker is cleaved—either enzymatically or via environmental triggers such as pH changes—releasing the cytotoxic payload into the intracellular space. Antigen expression, antibody affinity, binding site, and the intrinsic internalization dynamics of the antigen itself can impact the rate and degree of ADC internalization (4). Ongoing research is focused on optimizing these parameters to enhance the selective killing of tumor cells while sparing healthy tissues.
All FDA-approved ADCs, and most in clinical development, employ payloads directed against only a small set of molecular targets. Microtubule inhibitors such as auristatins and maytansinoids, disrupt cell division by inhibiting tubulin polymerization, ultimately leading to cell cycle arrest and apoptosis. Other common payloads, like topoisoperase I inhibitors and PBD dimers, induce DNA damage through multiple mechanisms, ultimately triggering cell death pathways. In addition, newer payloads are being explored that target specific oncogenic pathways or modulate the immune system, further diversifying the arsenal against cancer. The potency of these agents is balanced by the need for precise delivery—ensuring that even a small amount of the drug can exert cytotoxic effects while minimizing collateral damage to healthy tissues. Drug-to-antibody ratio (DAR) is a key parameter for optimization during ADC development and a critical quality attribute that must be monitored during manufacturing. DAR is most frequently measured by analytical methods, such as UV-Vis spectroscopy, LC-MS, hydrophobic interaction chromatography (HIC), and reversed-phase-HPLC. The relative utility of these methods depends on the nature of the conjugated payload and its linker.
In addition to the direct cytotoxic effects of ADC payloads, one of the most intriguing features of certain ADCs is their ability to induce a bystander killing effect. In this context, the released payload is not confined to the target cell alone but can diffuse into the surrounding tumor microenvironment, affecting neighboring cells that may not express the target antigen (5). This phenomenon is particularly beneficial in heterogeneous tumors where antigen expression can vary widely among cancer cells. The clinical importance of bystander killing can be readily seen in the dramatic response to T-DXd, which has bystander activity, relative to T-DM1, which does not (6). By broadening the spectrum of cell killing, bystander effects help in eradicating tumor cells that might otherwise escape direct targeting, thus enhancing the overall therapeutic impact of the ADC. Bystander killing effects of ADCs should be characterized during preclinical development, and may be an important criterion during linker optimization. Assays for bystander killing usually rely on mixtures cultures of differentially labeled antigen positive and negative cell lines. Specific depletion of the antigen-negative cells is monitored via low-throughput single-cell methods such as flow cytometry and imaging. A key consideration of this approach is that different cell lines often have very different susceptibilities to payload.
At the 2025 BEBPA US Bioassay conference, two talks were presented about ADCs. The first talk entitled “Roadmap for Bioassay Development for Antibody-Drug Conjugate” presented by Xuemin Wang, Associate Director of WuXi Biologics. Dr. Wang highlighted that market analyst, Biomedtracker, projected the global ADC market to grow from nearly 14 billion USD in 2025 to approximately 28 billion USD in the coming years, as one example of the importance of the ADC class of therapeutics. Dr. Wang then presented an ADC bioassay development roadmap. Four discrete steps were outlined including:
- Pilot Run – development of a prototype assay
- Development – further development of a QC friendly assay
- Robustness – refinement of critical reagents and qualification ready assay
- Qualification – qualified assay ready for GMP release and sample testing.
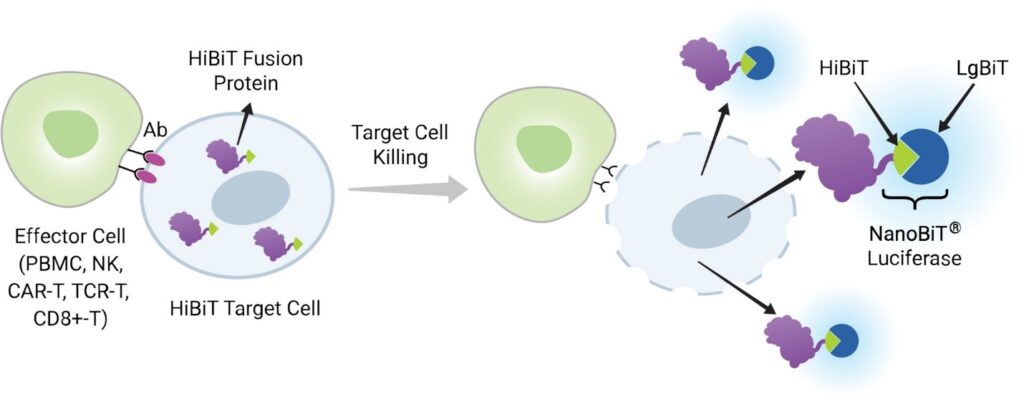
The second talk entitled “Novel Bioluminescent Tools for the Assessment of ADC internalization, Bystander Killing, and Fc Function” was presented by Dr. Julia Gilden, Senior Research Scientist of Promega Corporation. Dr. Gilden discussed a suite of technologies for the measurement of the biochemical and cellular events associated with ADC activity, which focused on the HiBiT Target Cell Killing (TCK) Platform (Figure 2) to measure bystander killing. HiBiT is an 11 amino acid peptide that binds the Large BiT protein with picomolar affinity, forming a bright, luminescent enzyme. HiBiT TCK cells express HiBiT as an intracellular fusion protein and when cells are lysed, HiBiT is released into the extracellular space and can complement Large BiT in the detection reagent. This results in a luminescent signal proportional to the number of lysed target cells. The HiBiT TCK platform was designed to specifically measure target cell death in mixed culture, enabling co-culture of cells that express an ADC target antigen, with HiBiT reporter cells that do not. This set-up allows for specific measurement of bystander killing. By using CRISPR to knock out antigens of interest, direct cytotoxicity and bystander effects can be quantified in the same cell background.
In summary, antibody–drug conjugates stand at the nexus of targeted therapy and precision medicine, offering a versatile platform for the treatment of cancer. With their intricate design, encompassing specific antibodies, carefully engineered linkers, and potent payloads, ADCs employ a diverse range of mechanisms—from immune effector engagement to bystander killing—to combat tumor cells effectively. The complexity of these molecules and their MOAs requires that a wide variety of analytical techniques and assay technologies be employed to fully characterize their structures and functions. Sustained innovation across ADC design, manufacturing, and assay technology will be essential to fully unlock the therapeutic potential of these multifaceted drugs.
- Crescioli, S., Kaplon, H., Wang, L., Visweswaraiah, J., Kapoor, V. and Reichert, J.M. (2025) Antibodies to watch in 2025. MAbs, 17, 2443538.
- Hoffmann, R.M., Coumbe, B.G.T., Josephs, D.H., Mele, S., Ilieva, K.M., Cheung, A., Tutt, A.N., Spicer, J.F., Thurston, D.E., Crescioli, S. and Karagiannis, S.N. (2018) Antibody structure and engineering considerations for the design and function of Antibody Drug Conjugates (ADCs). Oncoimmunology, 7, e1395127.
- Junttila, T.T., Li, G., Parsons, K., Phillips, G.L. and Sliwkowski, M.X. (2011) Trastuzumab-DM1 (T-DM1) retains all the mechanisms of action of trastuzumab and efficiently inhibits growth of lapatinib insensitive breast cancer. Breast Cancer Res Treat, 128, 347-356.
- Du, X., Beers, R., Fitzgerald, D.J. and Pastan, I. (2008) Differential cellular internalization of anti-CD19 and -CD22 immunotoxins results in different cytotoxic activity. Cancer Res, 68, 6300-6305.
- Kovtun, Y.V., Audette, C.A., Ye, Y., Xie, H., Ruberti, M.F., Phinney, S.J., Leece, B.A., Chittenden, T., Blattler, W.A. and Goldmacher, V.S. (2006) Antibody-drug conjugates designed to eradicate tumors with homogeneous and heterogeneous expression of the target antigen. Cancer Res, 66, 3214-3221.
- Hurvitz, S.A., Hegg, R., Chung, W.P., Im, S.A., Jacot, W., Ganju, V., Chiu, J.W.Y., Xu, B., Hamilton, E., Madhusudan, S. et al. (2023) Trastuzumab deruxtecan versus trastuzumab emtansine in patients with HER2-positive metastatic breast cancer: updated results from DESTINY-Breast03, a randomised, open-label, phase 3 trial. Lancet, 401, 105-117.

About The Author: Julia Gilden
Julia Gilden is a Senior Scientist at Promega Corp. Her group specializes in the development of bioassays for immuno-oncology applications, with a special focus on cell therapies and the use of primary human cells. She received her PhD in Biomedical Sciences with a focus in immunology from University of California, San Francisco and completed postdoctoral training in cell biology and parasitology at the University of Wisconsin Madison.

About The Author: Kai Hillman
Kai Hillman is an Associate Product Marketing Manager at Promega Corporation, where he specializes in the growth and engagement of cutting-edge bioassay solutions for biosimilars, vaccines, antibody-drug conjugates, and cell and gene therapies. Prior to joining Promega, he earned his PhD studying host-microbe interactions from the University of Wisconsin-Madison.
